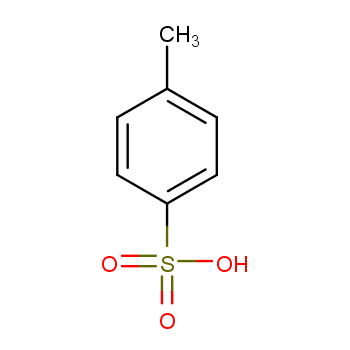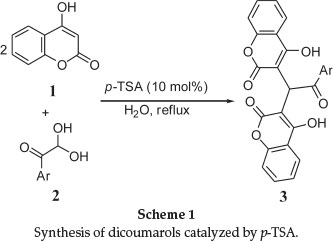
A facile and practical p-Toluenesulfonic acid catalyzed route to dicoumarols containing an Aroyl group

p-Toluenesulfonic acid monohydrate, 99%, extra pure, Thermo Scientific Chemicals, Quantity: 5 g | Fisher Scientific

p-Toluenesulfonic acid doped polystyrene (PS-PTSA): solvent-free microwave assisted cross-coupling-cyclization–oxidation to build one-pot diversely functionalized pyrrole from aldehyde, amine, active methylene, and nitroalkane - ScienceDirect

p-TSA@nano SiO2 as a New and Efficient Nanocatalyst for the One-Pot Multi-component Synthesis of Some Novel 1-Amidoalkyl-2-Naphthols Under Solvent-Free Conditions | SpringerLink

PTSA.H2O‐Catalyzed Reaction of 3‐Aminocoumarins and Phenylacetaldehydes: A Route to Access Various Pyrido(2,3‐c)coumarin Derivatives - Belal - 2017 - ChemistrySelect - Wiley Online Library
![One-Pot Three Component Domino Reaction for the Synthesis of Novel Isoxazolo[2,3-c][1,3,5]Thiadiazepin-2-Ones Catalyzed by PTSA—A Green Chemistry Approach One-Pot Three Component Domino Reaction for the Synthesis of Novel Isoxazolo[2,3-c][1,3,5]Thiadiazepin-2-Ones Catalyzed by PTSA—A Green Chemistry Approach](http://html.scirp.org/file/1-5500165x7.png)
One-Pot Three Component Domino Reaction for the Synthesis of Novel Isoxazolo[2,3-c][1,3,5]Thiadiazepin-2-Ones Catalyzed by PTSA—A Green Chemistry Approach

6192-52-5 | 4-Methylbenzenesulfonic Acid Monohydrate | 4-Methylbenzenesulfonic Acid Monohydrate; 4-Toluenesulfonic Acid Monohydrate; Tosic Acid Monohydrate; PTSA Monohydrate; p-Toluenesulfonic Acid Monohydrate; TsOH; p-tsa; Lisinopril EP Impurity B ...
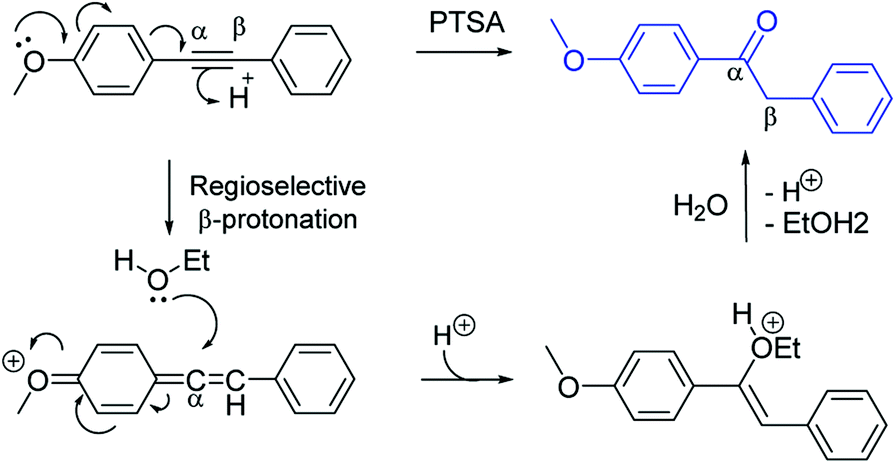
PtO 2 /PTSA system catalyzed regioselective hydration of internal arylalkynes bearing electron withdrawing groups - RSC Advances (RSC Publishing) DOI:10.1039/C8RA00564H

p-Toluenesulfonic acid, polymer-bound Macroporous, 30-60 mesh, extent of labeling: 2.0-3.0 mmol/g loading | Sigma-Aldrich

PTSA-Catalyzed Reaction of Alkyl/Aryl Methyl Ketones with Aliphatic Alcohols in the Presence of Selenium Dioxide: A Protocol for the Generation of an α-Ketoacetals Library | ACS Omega
![Para toluenesulfonic acid-catalyzed one-pot, three-component synthesis of benzo[5,6]chromeno[3,2-c]quinoline compounds in aqueous medium Para toluenesulfonic acid-catalyzed one-pot, three-component synthesis of benzo[5,6]chromeno[3,2-c]quinoline compounds in aqueous medium](https://www.degruyter.com/document/doi/10.1515/hc-2020-0128/asset/graphic/j_hc-2020-0128_fig_002.jpg)
Para toluenesulfonic acid-catalyzed one-pot, three-component synthesis of benzo[5,6]chromeno[3,2-c]quinoline compounds in aqueous medium