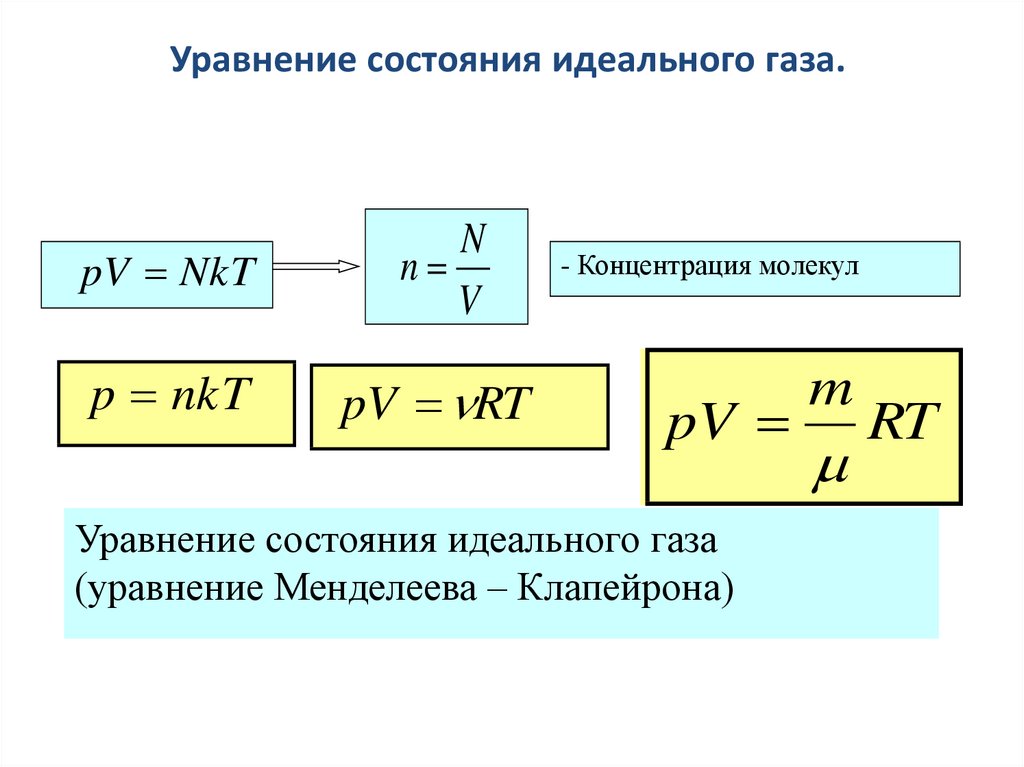
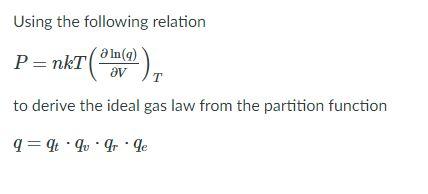SOLVED: Formula to predict the particles in the container is PV-NkT N= PVIkT Given P-47.3 atm V= 10.4 nm = 04x 10-26 T=300 K= 1.38x10-23 N= 2 (47.3)(1.04x 10-26 N = (1.38x10-23)(300) N= 532 mol
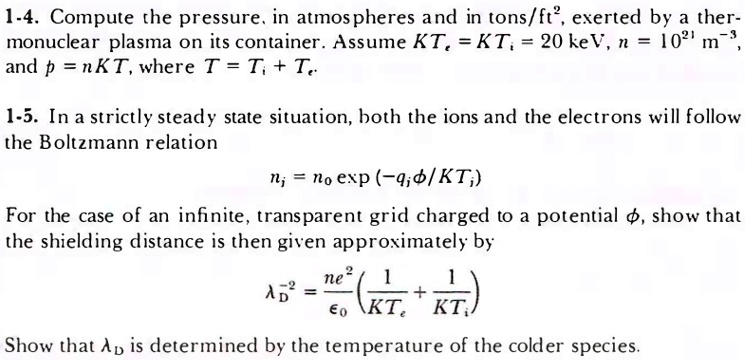
SOLVED: 1-4. Compute the pressure: in atmospheres and in tons/ft?, exerted by a ther monuclear plasma on its container Assume KT, KT; 20 keV, 102' and p nKT, where T = T; +
Skewed Distribution of Circulating Activated Natural Killer T (NKT) Cells in Patients with Common Variable Immunodeficiency Disorders (CVID) | PLOS ONE

METR February Review State variables: p, ρ, T Pressure Temperature Equation of state: p = NkT/V = ρ R d T Virtual temperature T v = T (1. - ppt download
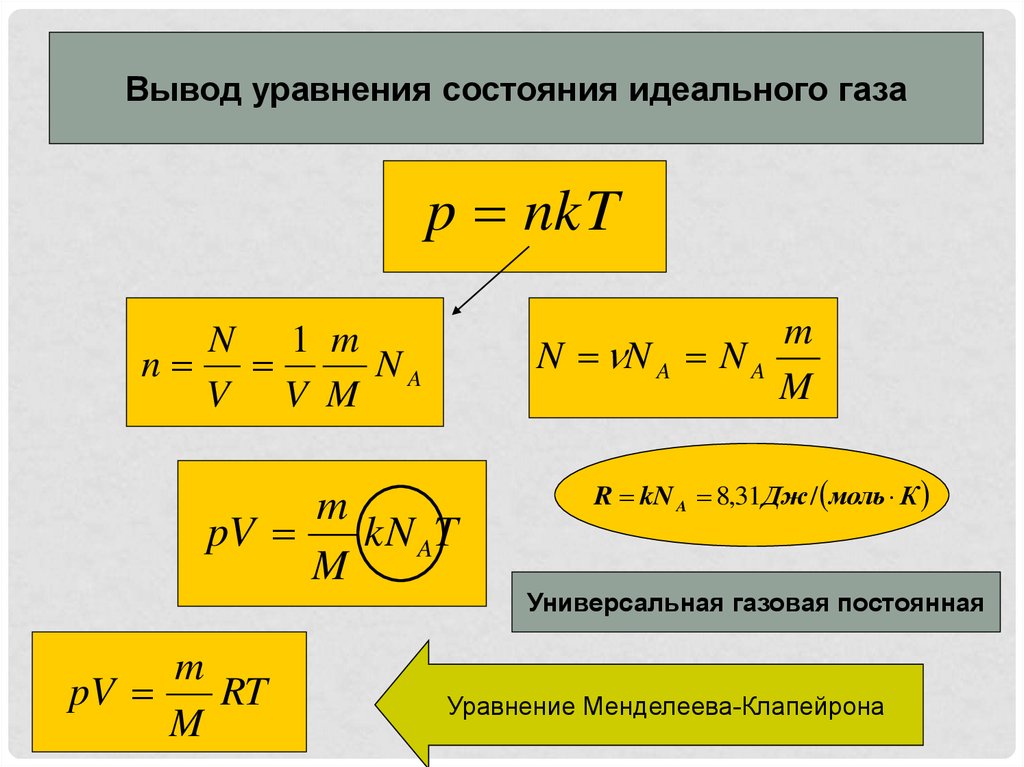
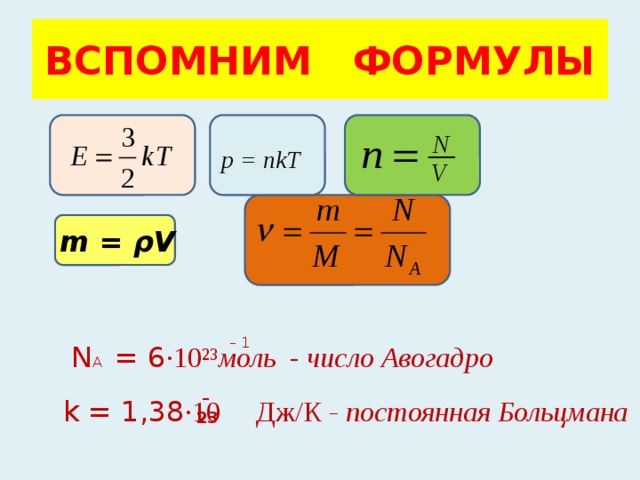
NkT PV = nRT PV = Pa pressure P = m volume V = moles n particles N # # = = kelvin e temperatur T = KJ x Const Boltzmann k / 1038