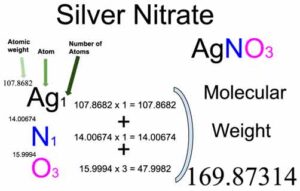
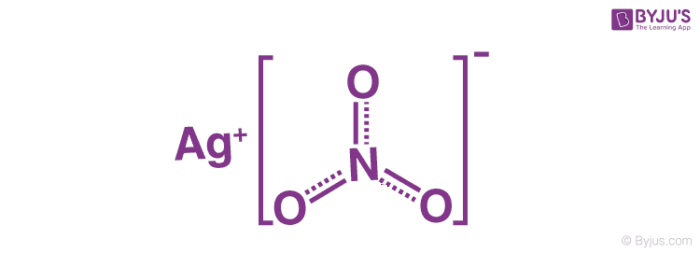
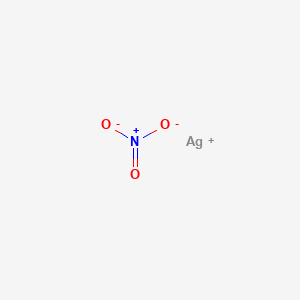
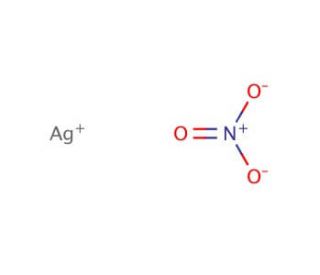
1) BaCl2 i AgNO3; 2) MgI₂ i AgNO3; 3) CaBr₂ i AgNO3; 4) Na3PO4 i AgNO3; 5) Ba(OH)₂ i H₂SO4; 6) - Школьные Знания.com

N-Halosuccinimide/AgNO3-Efficient Reagent Systems for One-Step Synthesis of 2-Haloglycals from Glycals: Application in the Synthesis of 2C-Branched Sugars via Heck Coupling Reactions | Organic Letters
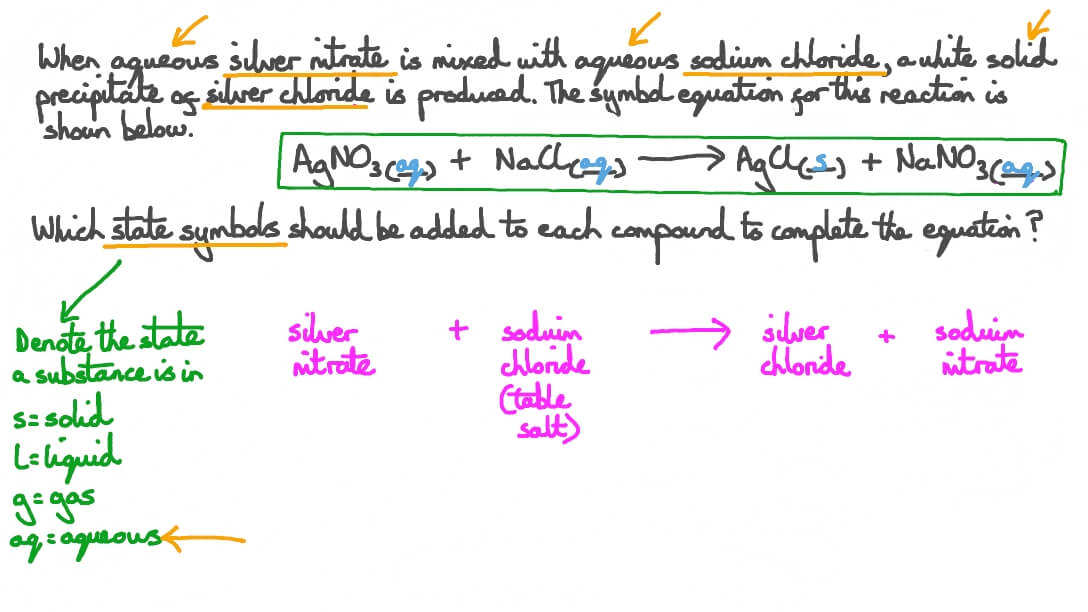
Question Video: Symbol Equation for the Reaction of the Precipitation of Agno3 with State Symbols | Nagwa

Thermo Scientific Chemicals Silver nitrate, ACS, 99.9+% (metals basis), Thermo Scientific Chemicals | Fisher Scientific
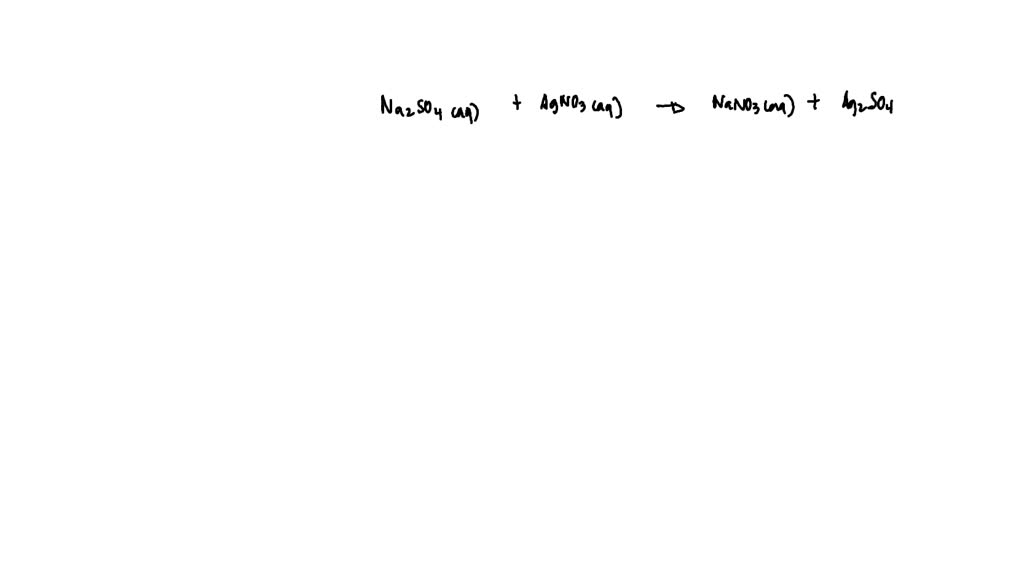
SOLVED: Which of the following solutions, when added to a solution of Na2SO4 would form a precipitate?I. AgNO3 (aq) II. KNO3 (aq) III. CaCl2 (aq)