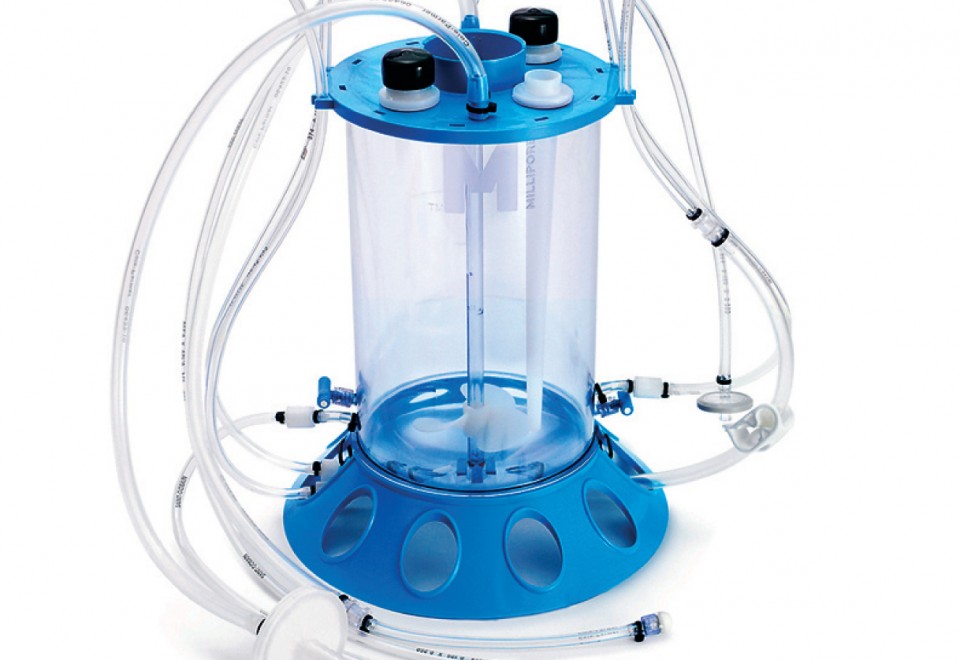
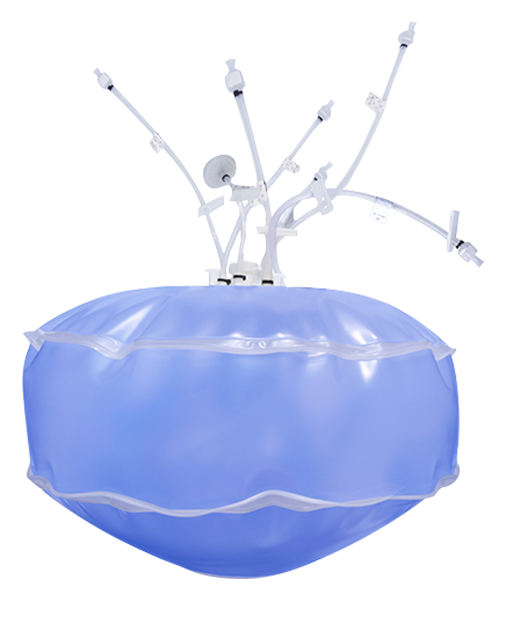
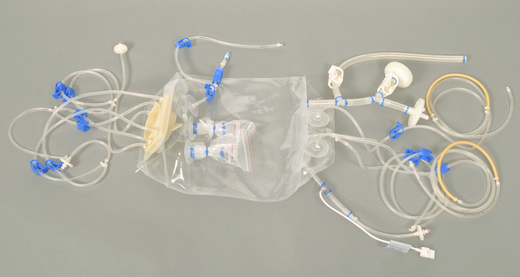
Thermo Scientific HyPerforma Single-Use Bioreactor TK, BioProcess Containers from Thermo Fisher Scientific - Product Description and Details

Expert Reverse Engineering Elicits Technical Data Necessary for Certification of Single-Use Bioreactor Systems | Accelerated Machine Design & Engineering LLC

Sartorius Stedim Biotech Launches New Single-use Bag Family Flexsafe - 2014 - Wiley Analytical Science

Pressure Decay Method for Postinstallation Single-Use Bioreactor Bag TestingBioProcess International

Development and Qualification of a Scalable, Disposable Bioreactor for GMP-Compliant Cell CultureBioProcess International